August 26, 2015 / Kyushu University / Scientific Reports
Text/Wu Tingyao
The research team of Kuniyoshi Shimizu, an associate professor at the Institute of Agricultural Sciences of Kyushu University in Japan, confirmed that 31 triterpenoids isolated from the fruiting body of Ganoderma inhibit the neuraminidase of five influenza A viruses to varying degrees, among which there are two triterpenoids even suitable for development as anti-influenza drugs. The research results were published in “Scientific Reports” under the “Nature” publishing group at the end of August 2015.
Neuraminidase is one of the two proteins protruding on the surface of influenza A viruses. Each influenza virus has about one hundred of these proteases. When the virus invades the cell and uses the material in the cell to replicate new virus particles, neuraminidase is needed for the new virus particles to break away from the cell and further infect other cells. Therefore, when neuraminidase loses its activity, the new virus will be locked in the cell and cannot escape, the threat to the host will be reduced, and the disease can be controlled. The commonly used oseltamivir (Tamiflu) in clinical practice is to use this principle to prevent the proliferation and spread of the virus.
According to research conducted by Kuniyoshi Shimizu, at a concentration of 200 μM, these Ganoderma triterpenoids inhibited the activity of H1N1, H5N1, H7N9 and two resistant mutant strains NA (H1N1, N295S) and NA (H3N2, E119V) to varying degrees. On the whole, the inhibitory effect on neuraminidase of N1 type (especially H5N1) is the best, and the inhibitory effect on neuraminidase of H7N9 is the worst. Among these triterpenoids, ganoderic acid TQ and ganoderic acid TR showed the highest levels of inhibition, and the effects of these two compounds ranged from 55.4% to 96.5% inhibition for different NA subtypes.
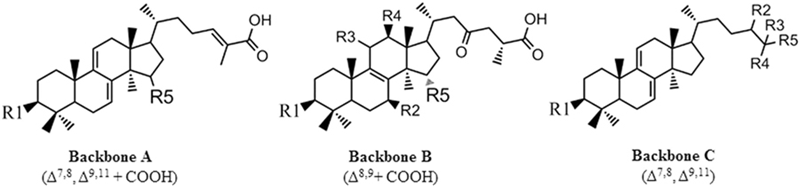
Further analysis of the structure-activity relationship of these triterpenoids revealed that the triterpenoids, which have a better inhibitory effect on N1 neuraminidase, have the main structure of “tetracyclic triterpenoids with two double bonds, a branch as a carboxylic group, and an oxygen-containing group at the R5 site” (Backbone A in the figure below). If the main structure is the other two (Backbone B and C in the figure below), the effect will be poor.
(Source/Sci Rep. 2015 Aug 26;5:13194.)
In silico docking is used to simulate the interaction of ganoderic acids (T-Q and TR) and neuraminidases (H1N1 and H5N1). As a result, it was found that both ganoderic acids and Tamiflu were able to directly bind to the active area of neuraminidase. This active area is composed of several amino acid residues. Ganoderma acids TQ and TR will bind to the two amino acid residues Arg292 and Glu119. Tamiflu has another option but can also make neuraminidase ineffective.
Compared with inhibiting other proteins on the influenza virus (such as the M2 protein, which opens the virus shell at the moment the virus binds to the host cell and sends the viral genes to the cell), neuraminidase inhibitors are currently recognized as effective and less resistant influenza treatment drugs. Therefore, researchers believe that ganoderic acids T-Q and TR, which are similar to but not the same in the mechanism of Tamiflu, have the opportunity to be used as a new generation of anti-influenza drugs or design references.
However, there is a prerequisite for the drug to be used as an anti-influenza drug, that is, the drug must effectively inhibit the reproduction of the virus without harming the cells infected by the virus. However, in experiments on cells infected with live viruses and breast cancer cell lines (MCF-7), it was found that when the researchers used these two types of ganoderic acids alone, they had doubts about high cytotoxicity, but they also found another kind of Ganoderma triterpenoid, ganoderol B, has an inhibitory effect on H5N1 (but the inhibitory effect is poor), but it is not cytotoxic. Therefore, researchers believe that how to improve the safety of ganoderic acids T-Q and TR through modification of the chemical structure while still retaining their inhibition of neuraminidase activity must be carefully considered.
[Source] Zhu Q, et al. Inhibition of neuraminidase by Ganoderma triterpenoids and implications for neuraminidase inhibitor design. Sci Rep. 2015 Aug 26;5:13194. doi: 10.1038/srep13194.
END
About the author/ Ms. Wu Tingyao
Wu Tingyao has been reporting on first-hand Ganoderma information since 1999. She is the author of Healing with Ganoderma (published in The People’s Medical Publishing House in April 2017).
★ This article is published under the exclusive authorization of the author. ★ The above works cannot be reproduced, excerpted or used in other ways without the authorization of the author. ★ For violations of the above statement, the author will pursue relevant legal responsibilities. ★ The original text of this article was written in Chinese by Wu Tingyao and translated into English by Alfred Liu. If there is any discrepancy between the translation (English) and the original (Chinese), the original Chinese shall prevail. If readers have any questions, please contact the original author, Ms. Wu Tingyao.
Post time: Sep-16-2021